The pharmaceutical industry built a $15 billion antidepressant market on serotonin as a brain chemical. The gut, which makes 95% of it, was not consulted.
Approximately 90–95% of the body's serotonin is produced in the gastrointestinal tract, not the brain, by specialized gut cells and bacteria that respond directly to what you eat. Gut-derived serotonin communicates with the brain's own serotonin system via the vagus nerve — a bidirectional highway where 80–90% of signals travel upward, from gut to brain. Dietary fiber, fermented foods, and tryptophan-rich meals directly influence the conditions for this serotonin production.
The most consequential correction in modern neuroscience did not make headlines. It appeared in a 2015 paper in Cell, confirmed by dozens of studies since, and still has not reached most people who eat food every day: roughly 90 to 95 percent of the body's serotonin is made in the gut, not the brain. It is made by specialized cells lining the intestinal wall, with the help of specific gut bacteria, from a single amino acid you can only get through food. The brain's serotonin system — the one pharmaceuticals target — is a downstream beneficiary of gut chemistry. And gut chemistry responds, within hours, to what you eat.
The Organ Doing the Work Nobody Credits
The gut has its own nervous system — roughly 500 million neurons embedded in the gastrointestinal lining, from the esophagus to the rectum. Gastroenterologists have long called this the enteric nervous system, informally the "second brain." That label undersells it. In one critical respect, it is not second at all.
Scattered throughout the gut wall are enterochromaffin (EC) cells — specialized sensory cells that detect mechanical pressure, nutrients, and microbial metabolites passing through the intestine. When they sense these stimuli, they do something the brain cannot: they manufacture serotonin from scratch. The raw material is tryptophan, an essential amino acid sourced entirely from dietary protein. The enzyme they use is tryptophan hydroxylase 1 (TPH1), which converts tryptophan into serotonin. The gut's serotonin does not cross the blood-brain barrier — but that is not where the story ends.
Enterochromaffin cells detect both mechanical stimuli (food moving through the gut) and chemical signals from microbial metabolites. In response, they release serotonin into the gut wall. This serotonin activates sensory neurons projecting to the vagus nerve, which carries the signal up to the brainstem — specifically to the nucleus tractus solitarius and, from there, to the dorsal raphe nucleus, which houses most of the brain's own serotonin-producing neurons. The gut does not just make serotonin. It tells the brain's serotonin system what to do.
“Colonization by a consortium of spore-forming bacteria from human microbiota significantly elevated colonic and blood serotonin levels, establishing that the gut microbiota plays a key regulatory role in intestinal EC cell serotonin production.”
The Vagus Nerve: Most Diagrams Draw It Backwards
Standard anatomy diagrams show the vagus nerve as a cable running from the brain down to the gut — implying the brain issues instructions. The actual fiber count tells a different story: approximately 80–90% of vagal nerve fibers are afferent — meaning they carry signals from the gut to the brain, not the other way around. The gut is not passively receiving commands. It is the one doing most of the reporting.
When EC cells release serotonin in response to a meal, that serotonin activates vagal afferent neurons. Those neurons transmit signals up to the nucleus tractus solitarius in the brainstem, which feeds into the dorsal raphe nucleus — the cluster of neurons responsible for producing most of the brain's serotonin. The gut's chemical output directly shapes the tone of the brain's serotonergic activity. Not metaphorically. Through a documented anatomical pathway.
Researchers studying oral SSRIs in mice found something unexpected: when gut-to-brain vagal signaling was surgically blocked, the antidepressant effects of the drugs were abolished — even though the drugs were still circulating systemically. The SSRIs were present. The brain was chemically exposed to them. But without the gut-brain signal, the mood effect disappeared. Part of what antidepressants may be doing is working through the gut-vagus-brain axis, not just acting on brain serotonin directly.
“Subdiaphragmatic vagotomy abolished the antidepressive effects of oral SSRI treatment, suggesting that vagus nerve dependent gut-brain signaling contributes substantially to the antidepressive effects of oral SSRIs.”
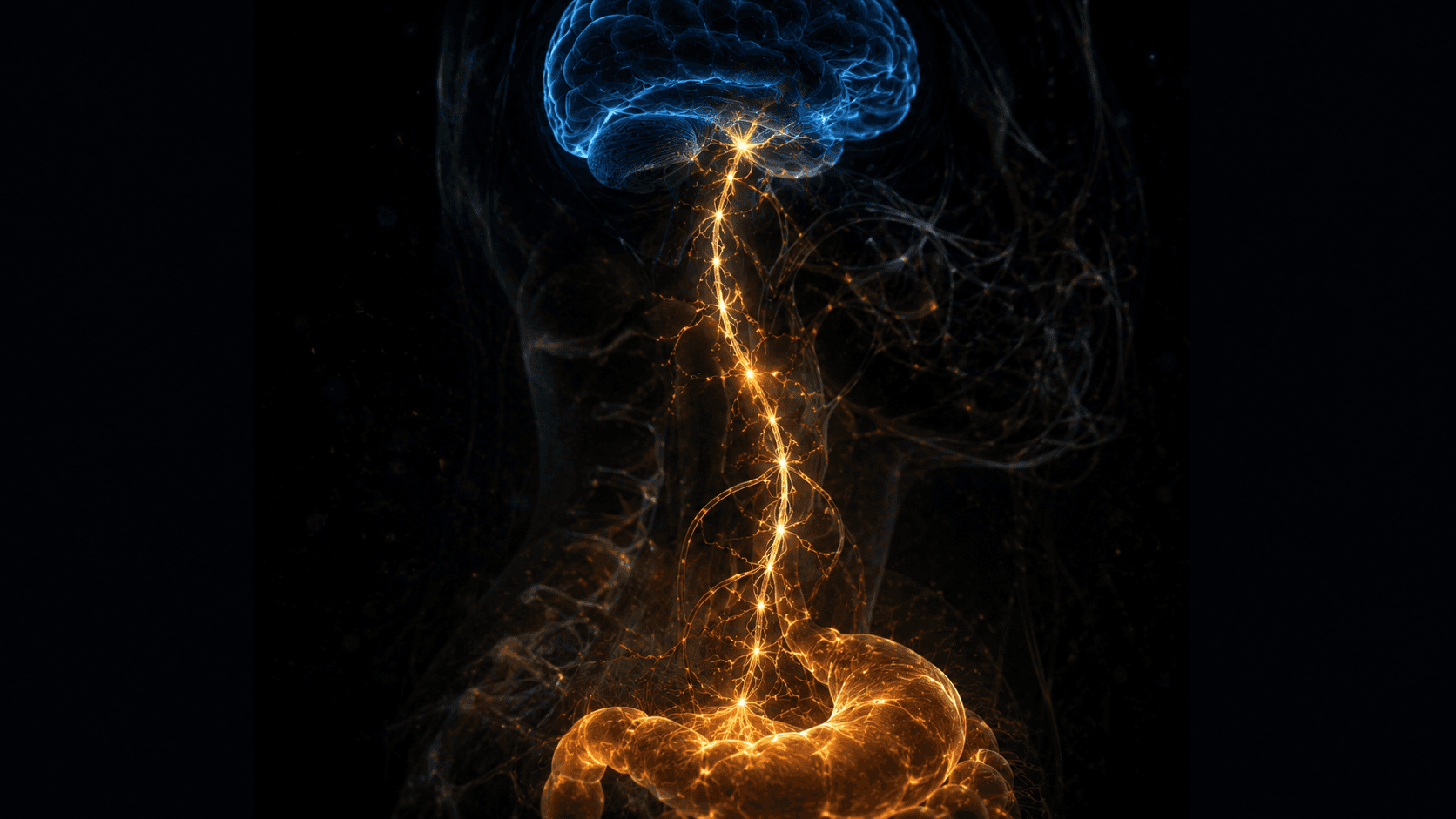
The Bacteria That Actually Build Your Mood Chemistry
The relationship between gut bacteria and serotonin is not indirect. Specific bacterial species produce serotonin themselves. A 2025 study published in Cell Reports identified two human gut bacteria — Limosilactobacillus mucosae and Ligilactobacillus ruminis — that synthesize bioactive serotonin via 5-HTP decarboxylation. Neither species could do it alone. The production required both species working in community — meaning microbial diversity, not just the presence of one strain, determines whether this pathway is active.
Even beyond direct serotonin production, gut bacteria shape the conditions for serotonin synthesis through a metabolic cascade: bacteria ferment dietary fiber into short-chain fatty acids (SCFAs) — primarily butyrate, propionate, and acetate. SCFAs directly stimulate TPH1, the enzyme that converts tryptophan into serotonin in EC cells. The logic chain is blunt: fewer fiber-fermenting bacteria means fewer SCFAs, which means less TPH1 activity, which means less serotonin synthesis.
“We identify a consortium of the human gut bacteria Limosilactobacillus mucosae and Ligilactobacillus ruminis that synthesizes serotonin in vitro by decarboxylation of 5-hydroxytryptophan and elevates fecal serotonin levels and colonic neuronal density.”

Tryptophan: The One Input Your Body Cannot Manufacture
The entire serotonin chain begins with tryptophan. The body cannot make it. Every molecule of tryptophan in your gut right now arrived through food — and once it arrives, it faces a choice: the gut's EC cells can convert it into serotonin via the TPH1 pathway, or the liver can convert it into kynurenine via the IDO pathway. Which direction it goes depends heavily on the state of the gut microbiome.
A healthy, diverse microbiome promotes the serotonin route. A depleted microbiome — reduced by antibiotics, chronic low fiber intake, or regular ultra-processed food consumption — shifts tryptophan metabolism toward the kynurenine pathway. Kynurenine metabolites include quinolinic acid, associated with neuroinflammation in animal models. Same amino acid. Radically different destination. The routing decision is made, in large part, by the bacteria present in your gut at the moment of digestion.
| Food | Tryptophan (mg / 100g) | Why It Matters |
|---|---|---|
| Pumpkin seeds | 576 | Highest plant source; also provides zinc and magnesium |
| Turkey (cooked) | 340 | Classic source; B6 supports conversion pathway |
| Cheddar cheese | 320 | High concentration; pairs well with carbs for uptake |
| Oats (dry) | 182 | Adds beta-glucan fiber — feeds SCFA-producing bacteria |
| Walnuts | 170 | Omega-3 reduces gut inflammation; supports microbiome |
| Firm tofu | 159 | Reliable plant option; calcium co-factor |
| Eggs (whole) | 167 | Choline supports gut lining integrity |
| Banana (ripe) | 9 | Low tryptophan but high resistant starch — prebiotic effect |
Eating carbohydrates alongside tryptophan-rich foods improves serotonin's reach in the brain — not because carbs contain tryptophan, but because the insulin spike clears competing large neutral amino acids from the bloodstream, giving tryptophan preferential access to the blood-brain barrier transporter. A food synergy effect, not a supplement interaction.

What Ultra-Processed Food Does to This Entire System
Ultra-processed food is not simply low-nutrient food. Its effect on the gut serotonin system is structural. Emulsifiers like carboxymethylcellulose and polysorbate-80 — common in packaged foods — disrupt the mucus layer protecting the gut epithelium. Artificial sweeteners alter microbial composition, reducing populations of fiber-fermenting bacteria. The result: less SCFA production, less TPH1 activity, and less serotonin synthesis. Meanwhile, a disrupted gut lining allows bacterial endotoxins to leak into systemic circulation — intestinal permeability — driving low-grade inflammation that extends to the brain.
A 2024 analysis published in Neurology, following over 72,000 participants across three cohort studies, found that every 10% increase in ultra-processed food consumption was associated with a 16% higher risk of cognitive impairment. A separate 2025 study in npj Metabolic Health and Disease identified structural changes in feeding-related brain regions linked to high ultra-processed food intake, partially mediated by systemic inflammation. The pathway in both cases runs through the gut.
Diverse, fiber-fed microbiome
- High SCFA production from fiber fermentation
- Active TPH1 stimulation in EC cells
- Efficient tryptophan → serotonin routing
- Intact gut mucosal barrier
- Low systemic inflammation
- Robust vagal signaling to brainstem
Depleted, low-fiber microbiome
- Reduced SCFA output
- Suppressed EC cell serotonin activity
- Tryptophan shifted toward kynurenine pathway
- Increased intestinal permeability
- Elevated inflammatory markers
- Weakened gut-brain signaling
“Each 10% increase in ultra-processed food consumption was associated with a hazard ratio of 1.16 for cognitive impairment, with inflammation and gut-mediated metabolic pathways proposed as primary mediating mechanisms.”
The Fiber Gap: 15 Grams When We Need 38
The average adult in Western countries consumes roughly 15 grams of fiber per day. Dietary guidelines recommend 25–38 grams. That gap is not a minor nutritional shortfall — it represents a chronic underfeeding of the exact bacterial populations responsible for SCFA production and TPH1 stimulation. Most fiber discourse focuses on bowel regularity. The actual leverage point is higher up the chain: without adequate fiber fermentation, the biochemical conditions for gut serotonin synthesis are structurally compromised.
The most effective prebiotic fibers — those specifically fermented by Bifidobacterium and Lactobacillus into butyrate and propionate — are found in unglamorous foods: onions, garlic, leeks, asparagus, Jerusalem artichokes, raw oats, unripe bananas, and cooked-then-cooled potatoes. Not superfoods. The raw substrate for the microbial chemistry that supports serotonin biosynthesis. The supplement industry sells the destination without the infrastructure.
Fiber-fermenting bacteria begin responding to increased substrate. Temporary bloating is normal — a sign the microbiome is actively processing.
SCFA production measurably increases. EC cell TPH1 activity supported by rising butyrate levels. Gut motility begins to normalize.
Community-level shifts in microbial diversity begin. Bifidobacterium and Lactobacillus populations expand with consistent prebiotic intake.
Measurable changes in microbial profile and SCFA output. Improved gut lining integrity. Sustained serotonin synthesis conditions established.
What the Research Actually Suggests Doing
Eat tryptophan with carbohydrates at the same meal
Tryptophan competes at the blood-brain barrier with other large neutral amino acids. Eating carbohydrates alongside tryptophan-rich foods triggers insulin, which clears competing amino acids from circulation — giving tryptophan preferential access. Practical examples: oats with eggs, turkey with sweet potato, cheese with whole-grain bread. Meal architecture, not supplementation.
High impactAdd one prebiotic food daily — from food, not powder
The evidence for isolated prebiotic supplements is substantially weaker than for whole-food sources. Garlic, onions, leeks, asparagus, and unripe bananas deliver inulin and fructooligosaccharides that Bifidobacterium and Lactobacillus ferment into SCFAs. Consistency matters more than dose. A clove of garlic in cooking, half an onion in a meal — sufficient to begin shifting microbial composition over weeks.
High impactAdd fermented foods to an already fiber-rich diet
Yogurt with live cultures, kefir, sauerkraut, kimchi, and miso introduce live bacteria that can transiently modulate gut composition. Current evidence positions these foods as most effective when added to a fiber-rich dietary base — not as replacements for one. A 2022 Stanford trial found a high-fermented-food diet increased microbiome diversity and reduced inflammatory markers, but fiber intake remained essential as substrate.
High impactReduce ultra-processed food by frequency, not portion size
Disruption to gut microbiome composition from emulsifiers and artificial sweeteners accumulates with daily exposure rather than single-meal quantity. Reducing how many days per week ultra-processed food is a staple appears more mechanistically relevant — and more behaviorally sustainable — than attempting to reduce portion size while maintaining daily frequency.
High impactThe Part This Article Cannot Tell You
The research discussed here concerns mechanisms of serotonin synthesis and gut-brain signaling — not clinical outcomes in mood disorder treatment. Dietary patterns that support gut microbiome diversity may influence the conditions for serotonin biosynthesis, but this does not constitute evidence that food can treat, replace, or substitute for clinical intervention in depression, anxiety, or any other condition. If you are experiencing persistent low mood, disrupted sleep, appetite changes, or any mental health concern, consult a qualified physician or psychiatrist.
The Reason to Eat Differently
The standard argument for eating well is that it is healthy — a generic statement that explains nothing and motivates no one for long. The mechanistic argument is more specific and more honest: the gut lining is a serotonin factory. It runs on tryptophan, fiber, and a community of bacteria that require daily substrate to stay functional. When the factory runs well, it sends signals up the vagus nerve that directly influence the brain's own serotonergic tone. When it doesn't, those signals degrade.
The brain has been taking credit for a chemical it barely produces. The gut has been running the operation. Every meal is, among other things, a report filed to the factory. The brain reads it. It responds. And that chain of chemistry begins not in a pharmacy — but in what is on your plate.

Get the science delivered weekly
No fads. No supplement ads. The research explained the way it should be.
Join the newsletterGetClariSync Nutrition Desk
Editorial Research · Nutritional Science
The GetClariSync Nutrition Desk reviews research in nutritional biochemistry, metabolism, and dietary science. We read across the American Journal of Clinical Nutrition, the British Journal of Nutrition, the Journal of Nutrition, Nutrients, and Cochrane Reviews — and we are explicit about what the evidence shows and where it is weak. We do not promote restrictive diets, supplements, or single-food claims unsupported by replicated research. We are editorial researchers, not registered dietitians or physicians — please consult a qualified nutrition professional or your doctor before significant dietary changes, especially if you have a health condition, take medication, are pregnant, or are managing a chronic disease.